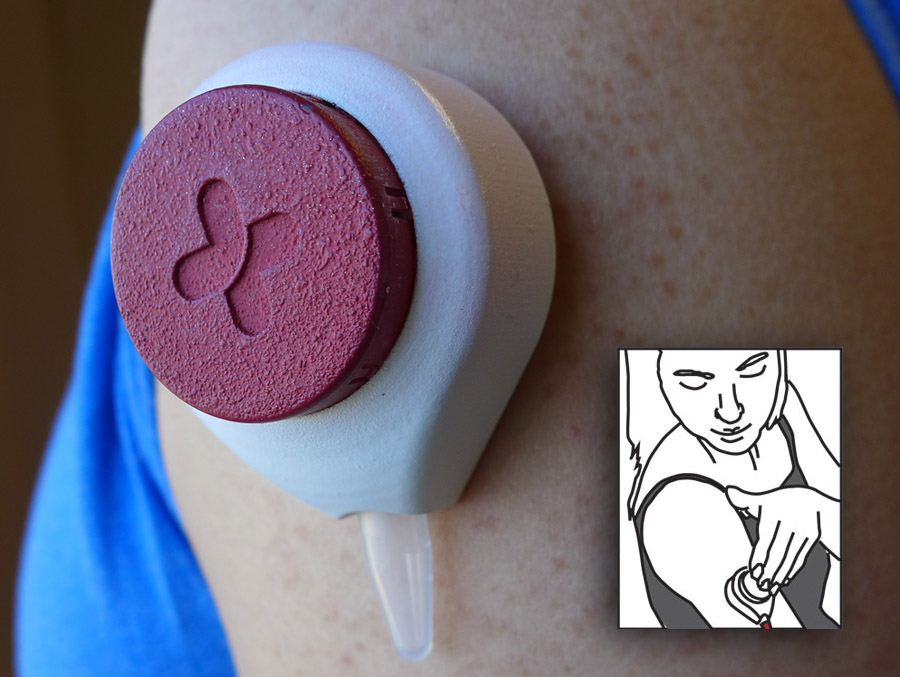
A new pain free Diabetes blood sugar test is being developed by Tasso Inc. The company is perfecting a device the size of a ping-pong ball that takes a small blood sample while being held against the skin for two minutes. Users report the procedure is nearly or completely painless. Those that suffer from Diabetes soon will have a new way to test their sugar levels without sticking themselves with a needle.
This test relies on a little vacuum in the unit enables a tiny sample of blood to flow into an attached sample vial. The blood sample can then either be analyzed on the spot (for diabetic blood sugar testing) or be sent to your laboratory for more advanced testing.
The organization has just received an added $3 million from the Defense Advanced Research Projects Agency (DARPA) to improve this device.
The technology relies on the powers that regulate the stream of fluid that is miniature flows. Surface tension keeps the blood in the channel however you hold the unit.
The firm has applied for a patent on the systems that create the capillary action to transfer blood toward the sample tube. Transferring the blood in open channels rather than conventional channels simply the process and cut the cost for the disposable device.
Work will be funded by the DARPA grant with businesses that produce blood preservatives. The next aim of the company is to stabilize the blood for a week at 140 degrees Fahrenheit for transportation to a lab. That will enable the creation to be utilized to sample in an extensive variety of places without needing expensive cold chain transfer.
Tasso Inc. is also creating a technique for choosing samples to diagnose HIV with funds from the National Institutes of Health. Now, these devices can take out about 0.15 of a cubic centimeter, enough to examine cholesterol, disease, cancer cells and blood sugar.
These devices could help people who dread blood draws, and prevent time consuming trips to laboratories for blood draws. If all goes well, the firm plans to release the product into the marketplace during 2016, and is going to send its application to the FDA by the end of 2015.